A growing global effort is underway to better understand the...
Read MoreAustralian Research: The REPEAT Trial Brings New Hope for PCD
Australia is playing a leading role in advancing care for people living with Primary Ciliary Dyskinesia (PCD) through the groundbreaking REPEAT Trial—an innovative research study led by Anne Chang. Catherine Kruljac, President and Founder of PCD Australia, was privileged to serve on the trial’s committee, contributing valuable patient advocacy insight and supporting the representation of the PCD community throughout the study.
The REPEAT Trial (Reducing Exacerbations in Primary Ciliary Dyskinesia) is a multi-centre, randomised controlled trial conducted across Australia, with sites in the Northern Territory, Queensland, New South Wales, Victoria and Western Australia. This nationally coordinated effort reflects a significant step forward in addressing the limited evidence base for treating PCD.
Why the REPEAT Trial Matters
PCD is a rare, lifelong condition that affects the body’s ability to clear mucus from the lungs, leading to chronic infections and progressive lung disease. Despite its impact, there have been very few large-scale clinical trials specifically focused on PCD, meaning treatment approaches are often adapted from other respiratory conditions.
The REPEAT Trial aims to change that.
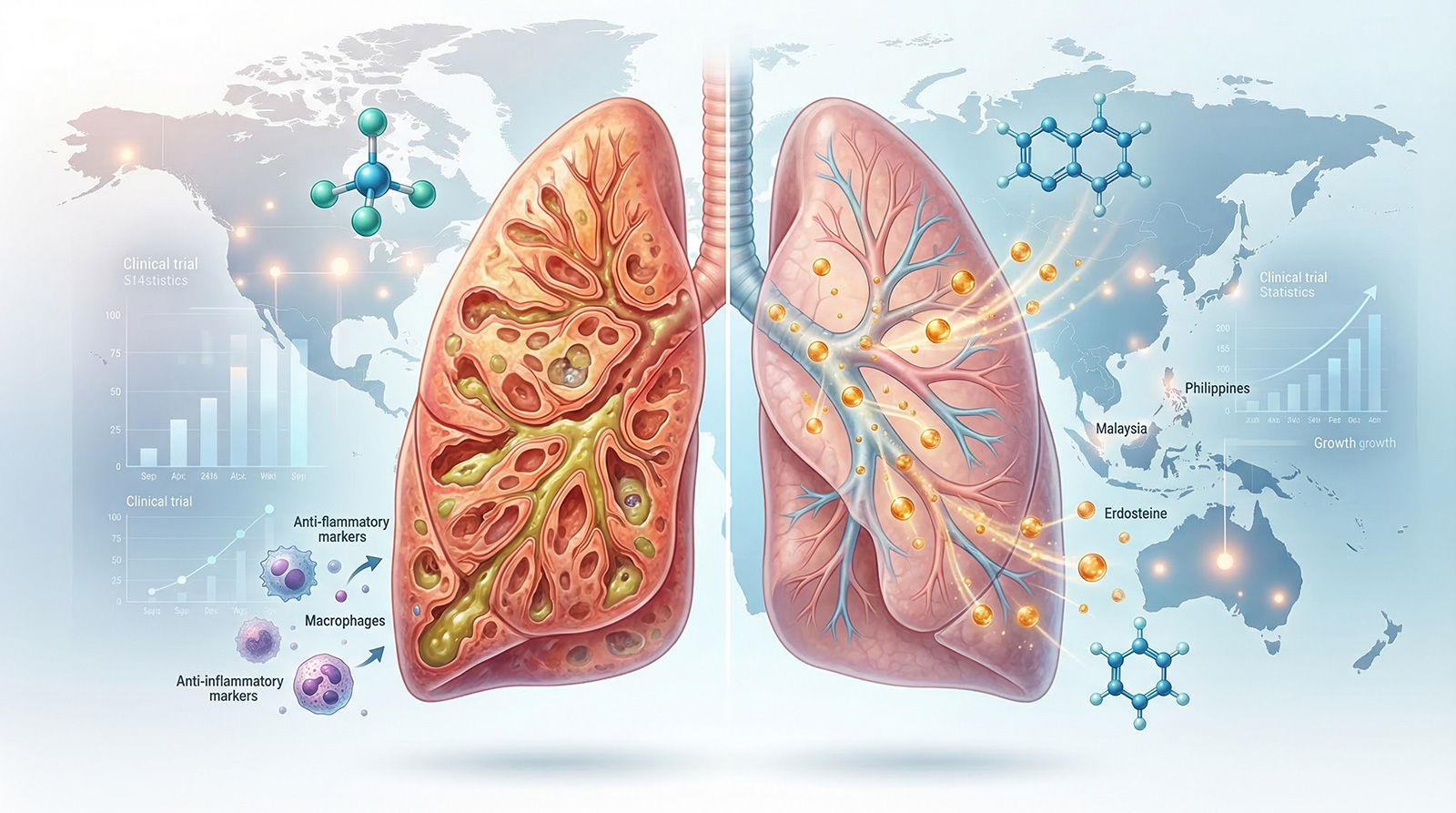
This study is investigating whether a combination of treatments—specifically the antibiotic azithromycin and the mucolytic medication Erdosteine—can reduce the frequency and severity of respiratory exacerbations in both children and adults with PCD over a 12-month period. (PubMed)
A National and Global Effort
Led by Professor Anne Chang, alongside leading researchers across Australia and international collaborators, the REPEAT Trial represents a powerful example of collaboration in rare disease research. It is funded through the Medical Research Future Fund and supported by key organisations including the Lung Foundation Australia and patient advocacy groups. (CRE in Respiratory Health)
Importantly, the trial includes approximately 100 participants from across the country, ensuring that outcomes reflect real-world experiences across diverse communities. (PubMed)
Potential Impact for the PCD Community
The goal of the REPEAT Trial is not only to reduce chest infections but also to improve long-term lung health, quality of life, and overall disease management. By preventing repeated exacerbations, the study has the potential to slow disease progression and reduce hospitalisations for people living with PCD. (CRE in Respiratory Health)
If successful, the findings could:
- Provide the first strong evidence for routine use of these medications in PCD
- Influence clinical guidelines both in Australia and internationally
- Improve access to effective, evidence-based treatments
The Role of the Community and PCD Australia
A key strength of the REPEAT Trial is the involvement of the PCD Australia and their community. Patients and families are central to the research, contributing not only through participation but by helping shape outcomes that matter most—such as quality of life and day-to-day wellbeing.
For PCD Australia, this trial represents a significant milestone. It highlights the importance of Australian-led research and ensures that local patients are part of global progress in understanding and treating PCD.
Looking Ahead
While results are still emerging, the REPEAT Trial signals a shift toward more targeted, evidence-based care for PCD. It brings hope that future treatments will be better tailored, more effective, and grounded in robust clinical research.
Through continued collaboration between researchers, clinicians, and the patient community, Australia is helping lead the way toward improved outcomes for everyone living with PCD.
Recent Posts
World Rare Disease Day from PCD Committee Member, and PCD Mother, Steph March
Stephanie March And Joel Taggart Are Urging Councils Across South...
Read MoreYou Don’t Know What You Know Until You Ask … So We Did
PCD Australia recently circulated a Listening Survey to gain a...
Read MoreLung Legends
We are proud to share that Lung Foundation Australia has...
Read More